Just wondering how stable everyone's alk is, especially those using the trident auto dosing or not.
Please post screen shots of your graphs and any tips you may have.
Please post screen shots of your graphs and any tips you may have.
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.

Would love to read it!I'm about to post a new thread that summarizes a question that came up in the KH director forum since it bears on what alkalinity stability really means (and perhaps whether people are being mypoic on it vs pH).
I'll post the link here when it is up.

Would love to read it!
Wow that’s amazing how perfect your ph swingsI see at max a .3 swing. Working to dose a little more at night to level that swing out.
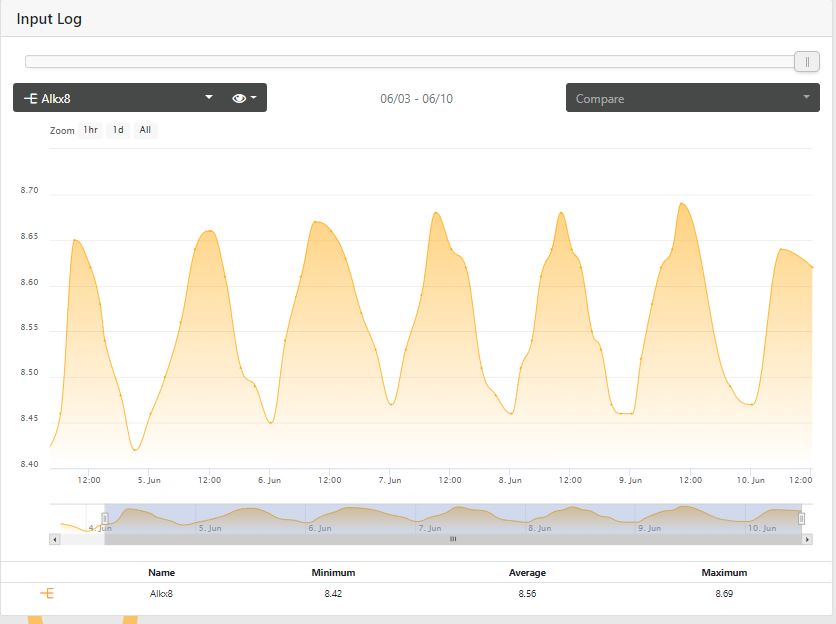

Wow that’s amazing how perfect your ph swings
Randy, when you say "...whether alk is the actual thing that is most usefully stabilized vs bicarbonate" I'm assuming you are referring to stabilizing pH (rather than alk) to stabilize the bicarbonate:carbonate ratio? If so, I've been wondering what you and others think of that idea of stabilizing pH. I've been working on a crude "experiment" where I have attempted to maintain a constant pH (of 8.3) using a GHL Controller and motorized ball valves to regulate airflow with different CO2 levels going to the skimmer. My plan was to start a thread on this but I have been reluctant because it turned out to be a long "read" and with less than ideal results due to some problems I ran into. But still, I am very curious about the pros and cons of maintaining a constant pH if that could be achieved.While how stable the alk is is an important consideration for users and potential users of this device, the more interesting thing (IMO) and the much harder thing to quantify is how the tank is different with more stable alk vs less stable alk, and even whether alk is the actual thing that is most usefully stabilized vs bicarbonate.

Randy, when you say "...whether alk is the actual thing that is most usefully stabilized vs bicarbonate" I'm assuming you are referring to stabilizing pH (rather than alk) to stabilize the bicarbonate:carbonate ratio? If so, I've been wondering what you and others think of that idea of stabilizing pH. I've been working on a crude "experiment" where I have attempted to maintain a constant pH (of 8.3) using a GHL Controller and motorized ball valves to regulate airflow with different CO2 levels going to the skimmer. My plan was to start a thread on this but I have been reluctant because it turned out to be a long "read" and with less than ideal results due to some problems I ran into. But still, I am very curious about the pros and cons of maintaining a constant pH if that could be achieved.
(Sorry if I briefly got off track from original post).

 www.reef2reef.com
www.reef2reef.com
