I’m planning on using the tropic Marin Trace A/K dosed separately, it’s way down there at the end of the original post.I don't see anything adding trace elements.
You need to do that separately or rely on regular water changes to top them up.
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
New DIY Two Part Recipes with Higher pH Boost
- Thread starter Randy Holmes-Farley
- Start date
- Tagged users None
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
My magnesium part and Balling Part C are somewhat redundant (both are mostly magnesium and contain a lot of sulfate) and I do not recommend both. Use balling C and then only use the mag part if testing suggests a need. The ratio won’t matter too much, but in that scenario use the stand alone mag ratio of 11:1.
The Balling C is used at the rate that BRS recommends for the soda ash recipe.
The Balling C is used at the rate that BRS recommends for the soda ash recipe.
Thanks randy, that was kind of my plan. Only dose the mag when it goes low since I have daily mag readings whether I want them or not with Trident.My magnesium part and Balling Part C are somewhat redundant (both are mostly magnesium and contain a lot of sulfate) and I do not recommend both. Use balling C and then only use the mag part if testing suggests a need. The ratio won’t matter too much, but in that scenario use the stand alone mag ratio of 11:1.
The Balling C is used at the rate that BRS recommends for the soda ash recipe.
the magnesium and sulfate portions were kind of confusing since your two part combined them but I’m not doing that, so I wanted to verify the ratios.
I appreciate you taking the time to look that over.
1) Soda Carbonate & Sodium hydroxide will have the same alkalinity boost when you follow Randy’s recipes.
2) If you are going to use Tropic Balling Part C, the magnesium mix will be slightly different:
I pulled the measurements and numbers right off of this page. I've read BRS uses Randy's recipe, but I don't know if this is specifically the one they got from him as it has Balling C incorporated in it.Why 2x?
Kalkwasser does NOT have a salinity imbalance effect. You would follow the mixing instructions of balling part C (91g/gal) and dose equal amounts of soda ash, calcium, and balling.
You can technically dose balling once a week, but it isn’t ideal.
Balling Part C is NOT a trace element additive. It is used to prevent skewing the salinity toward sodium and chloride.

Trace elements for your BRS Bulk 2-Part : The BRS / Tropic Marin Hybrid Balling
Learn how to mix and dose the BRS Bulk 2-Part / Tropic Marin Hybrid Balling Method
I basically want this recipe with Sodium Hydroxide instead of Soda Ash as they have listed.
You're saying their listed magnesium mix isn't ideal and I should do 933 grams Magnesium Chloride and 84.82 grams Magnesium Sulfate per Gallon for 11 to 1 ratio as needed and dose Balling C routinely?
Last edited:
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
You're saying their listed magnesium mix isn't ideal and I should do 933 grams Magnesium Chloride and 84.82 grams Magnesium Sulfate per Gallon for 11 to 1 ratio as needed and dose Balling C routinely?
The listed magnesium recipe is best if not using Balling Part C. If using Part C, the one you mentioned us vests if you need extra magnesium.
- Joined
- Jun 4, 2016
- Messages
- 1,002
- Reaction score
- 455
Read through all 37 pages. Reading comprehension and retention is now going to be tested as I embark on this new Sodium Hydroxide journey. lol.
I'm running a CaRX and biopellets in a energy efficient home that is likely too well sealed all contributing to the CO2 thus pH problem in my reef. I plan to back down the CaRX when starting the Part 2 dosing as to not overdose the alk. Question is to the Part C. I see the keeping balance part and potentially trace elements, but what are the things to be concerned about if running Part 2 Sodium Hydroxide alone in tandem with the CaRX with manual weekly trace element supplements? I saw salinity creep which I'm fine with scooping a bit of tank water out occasionally to let fresh ATO rebalance. No biggie.
I'm running a CaRX and biopellets in a energy efficient home that is likely too well sealed all contributing to the CO2 thus pH problem in my reef. I plan to back down the CaRX when starting the Part 2 dosing as to not overdose the alk. Question is to the Part C. I see the keeping balance part and potentially trace elements, but what are the things to be concerned about if running Part 2 Sodium Hydroxide alone in tandem with the CaRX with manual weekly trace element supplements? I saw salinity creep which I'm fine with scooping a bit of tank water out occasionally to let fresh ATO rebalance. No biggie.
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
Read through all 37 pages. Reading comprehension and retention is now going to be tested as I embark on this new Sodium Hydroxide journey. lol.
I'm running a CaRX and biopellets in a energy efficient home that is likely too well sealed all contributing to the CO2 thus pH problem in my reef. I plan to back down the CaRX when starting the Part 2 dosing as to not overdose the alk. Question is to the Part C. I see the keeping balance part and potentially trace elements, but what are the things to be concerned about if running Part 2 Sodium Hydroxide alone in tandem with the CaRX with manual weekly trace element supplements? I saw salinity creep which I'm fine with scooping a bit of tank water out occasionally to let fresh ATO rebalance. No biggie.
The issue is that using just calcium chloride and sodium (bicarbonate/carbonate/hydroxide) continually adds sodium and chloride.
Assuming you maintain salinity (you need to), then every time you correct the salinity downward, everything drops, not just trace elements. Potassium, sulfate, bromide and on and on. That's what Part C adds.
In a year of adding 1.1 dKH and no additions or water changes, everything except sodium and chloride (and calcum/alk and any impurities in the products) is depleted by a factor of 22.5%.
This recent post has more:

Unbalance Na/Cl-based products question
@Randy Holmes-Farley When does unbalanced Na/Cl-based dosing impact a reef tank? I've always tried to practice balance dosing to keep my parameters in line with natural or at least within acceptable ranges. To me balanced dosing = keeping the ionic concentrations. For instance, typical...
 www.reef2reef.com
www.reef2reef.com
- Joined
- Jun 4, 2016
- Messages
- 1,002
- Reaction score
- 455
Hi Randy,
Glad you raised the no water change position as that's exactly my camp thus I rely on dosing to replenish. i.e. weekly trace and other water supplements. If everything else drops not just trace, then dosing potassium, sulfate, bromide etc manually could keep it in balance?
I mixed up a test 1000ml at 1.5x (~110 g of NaOH to 1000 ml RODI) and started today. Doing only 2 ml/hour at the moment into a 180g. I'm still trying to figure out the balance between CaRX and NaOH dosing so starting very minimum just to introduce it into the system and establishing a baseline. I don't expect 48ml/daily into 180g is going to do much so I have time to adjust and react. The calculator shows that I need to add 347 ml of ESV Bicarbonate Syst Pt1 to raise 1 dKH in my 180g. My 48ml shouldn't have any impact but maybe to observe the pH rise. @Mindi in the first several pages of this thread seem to be heading the direction I'm looking for. I don't want/need any benefits for this solution except to boost pH since I'm running the CaRX
I've seen folks use kalk dosing and/or in a kalk stirrer to supplement their CaRX as a combo to help with pH. I'm assuming this NaOH dosing in place of kalk since it's more potent and I'm able to use less solution thus less refill if compared to saturated kalk traditional dosing.
Am I approaching this in the wrong way and I should just still to kalk dosing supplement? Accept the refill effort for saturated kalk in a doser!?
Glad you raised the no water change position as that's exactly my camp thus I rely on dosing to replenish. i.e. weekly trace and other water supplements. If everything else drops not just trace, then dosing potassium, sulfate, bromide etc manually could keep it in balance?
I mixed up a test 1000ml at 1.5x (~110 g of NaOH to 1000 ml RODI) and started today. Doing only 2 ml/hour at the moment into a 180g. I'm still trying to figure out the balance between CaRX and NaOH dosing so starting very minimum just to introduce it into the system and establishing a baseline. I don't expect 48ml/daily into 180g is going to do much so I have time to adjust and react. The calculator shows that I need to add 347 ml of ESV Bicarbonate Syst Pt1 to raise 1 dKH in my 180g. My 48ml shouldn't have any impact but maybe to observe the pH rise. @Mindi in the first several pages of this thread seem to be heading the direction I'm looking for. I don't want/need any benefits for this solution except to boost pH since I'm running the CaRX
I've seen folks use kalk dosing and/or in a kalk stirrer to supplement their CaRX as a combo to help with pH. I'm assuming this NaOH dosing in place of kalk since it's more potent and I'm able to use less solution thus less refill if compared to saturated kalk traditional dosing.
Am I approaching this in the wrong way and I should just still to kalk dosing supplement? Accept the refill effort for saturated kalk in a doser!?
Last edited:
- Joined
- Jun 4, 2016
- Messages
- 1,002
- Reaction score
- 455
Same, but not limited in dosing by evaporation rates.
Reread the earlier response when someone asked about the pH impact of sodium hydroxide vs kalk. Randy's response was same, however it appears there's a saturation limit with kalk where sodium hydroxide seems to be able to mix significantly more potentially. i.e. dose less for same results = less solution to refill?
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
Reread the earlier response when someone asked about the pH impact of sodium hydroxide vs kalk. Randy's response was same, however it appears there's a saturation limit with kalk where sodium hydroxide seems to be able to mix significantly more potentially. i.e. dose less for same results = less solution to refill?
Yes, the pH effect per unit of alk added is the same, but the volume of water needed with sodium hydroxide is much less.
- Joined
- Jun 4, 2016
- Messages
- 1,002
- Reaction score
- 455
Yes, the pH effect per unit of alk added is the same, but the volume of water needed with sodium hydroxide is much less.
That confirms it! =) I'll use sodium hydroxide as an alternative to kalk given the less volume of water needed thus ability to make it super potent.
This thread is about an alternative 2 part recipe with a higher pH. You've addressed earlier that one should doing both parts 1 and 2 to keep the balance. Sounds like without it one might address the imbalance somehow.
However I did overlook something which I can't believe I did. PART 2 recipe calls for sodium hydroxide + sodium sulfate. What if the sodium sulfate is omitted altogether and dose a sodium hydroxide only solution. Will it continue to increase kalk and pH again serving as a kalk alternative? I accidently missed the sodium sulfate and have been dosing without this ingredient in the recipe for half a day. pH seems to be elevated, but not sure of any other advise affect especially over time.
How important are the magnesium and sulfate components if weekly 10% water changed are done? I am talking both option when mixed with alk and cal or done as a true 3 part. Will weekly water changes keep everything in balance?
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
A water change cannot eliminate the problem, but certainly reduces it.How important are the magnesium and sulfate components if weekly 10% water changed are done? I am talking both option when mixed with alk and cal or done as a true 3 part. Will weekly water changes keep everything in balance?
I’d personally use Balling Part C for the third part. That works better than water changes for this purpose, and is likely cheaper than water changes anyway.
- Joined
- Oct 20, 2019
- Messages
- 75
- Reaction score
- 59
Currently dosing 220ml for a 150G tank, consumption is still increasing (roughly 2dKh° per day).
How high should I be concerned?
May I start to dose baking soda instead of sodium hydroxide let's say when over 8.5?
Probe has been just calibrated few days ago
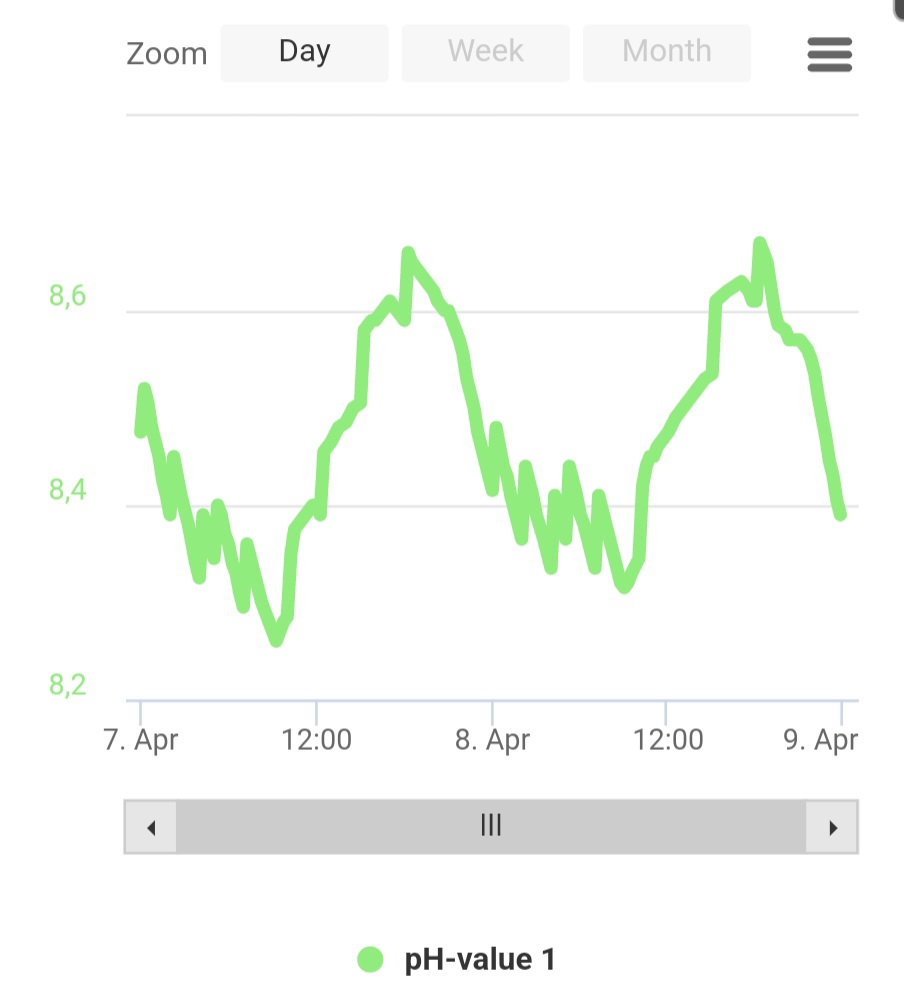
How high should I be concerned?
May I start to dose baking soda instead of sodium hydroxide let's say when over 8.5?
Probe has been just calibrated few days ago
Attachments
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
Seems ok, but demand will be higher at this higher pH.
I've switched to this recipe from the original and All for reef, for the PH boost, its been great.
Im wondering how strong I can make the solutions. so ESV is 1.5, but I'm looking to make a 2x strength. is 3x or 4x possible? what are the solubility limits
Im wondering how strong I can make the solutions. so ESV is 1.5, but I'm looking to make a 2x strength. is 3x or 4x possible? what are the solubility limits
- Joined
- Jul 8, 2017
- Messages
- 102
- Reaction score
- 64
I've been reading over these threads over the last few days. Thanks for the wealth of knowledge! I'd like to incorporate a 2-part solution that will raise my pH as high as possible to replace kalk in my ATO.
I saw this pH boost recipe in another thread. Could you help me to understand the difference in these two recipes, I'm having trouble combing through the threads to compare:
From the other thread:
Alk part
Add 283 grams of sodium hydroxide to 1 gallon of fresh water. It will get quite warm. Make sure it doesn't soften your container. This solution will contain about 1,900 meq/L of alkalinity (5,300 dKH). BE CAREFUL WITH THIS SOLUTION: IT HAS A pH ABOVE 14. Do not get it in your eyes or on your skin. Keep all reef chemicals, especially this alk part, in a way that children cannot access them.
Calcium part
Dissolve 500 grams (about 2 ½ cups) of calcium chloride dihydrate (such as Dowflake 77-80% calcium chloride or ESV calcium chloride; see below for substitutes and sources) in enough water to make 1 gallon of total volume. You can dissolve it in about ½ gallon of water, and then pour that into the 1 gallon container and fill it to the top with more freshwater. This solution has about 37,000 ppm calcium.
Magnesium part
Dissolve Epsom salts (magnesium sulfate heptahydrate (3 cups) and magnesium chloride hexahydrate (5 cups) in enough purified freshwater to make 1 gallon total volume. There will likely be a precipitate that forms even if you fully dissolve both ingredients separately. That precipitate is calcium sulfate (calcium as an impurity in the magnesium chloride and sulfate from the Epsom salts). It is fine and appropriate to dose the precipitate along with the remainder of the fluid by shaking it up before dosing.
VS the "Sodium hydroxide version" from post 1 in this thread.
Thank you!
I saw this pH boost recipe in another thread. Could you help me to understand the difference in these two recipes, I'm having trouble combing through the threads to compare:
From the other thread:
Alk part
Add 283 grams of sodium hydroxide to 1 gallon of fresh water. It will get quite warm. Make sure it doesn't soften your container. This solution will contain about 1,900 meq/L of alkalinity (5,300 dKH). BE CAREFUL WITH THIS SOLUTION: IT HAS A pH ABOVE 14. Do not get it in your eyes or on your skin. Keep all reef chemicals, especially this alk part, in a way that children cannot access them.
Calcium part
Dissolve 500 grams (about 2 ½ cups) of calcium chloride dihydrate (such as Dowflake 77-80% calcium chloride or ESV calcium chloride; see below for substitutes and sources) in enough water to make 1 gallon of total volume. You can dissolve it in about ½ gallon of water, and then pour that into the 1 gallon container and fill it to the top with more freshwater. This solution has about 37,000 ppm calcium.
Magnesium part
Dissolve Epsom salts (magnesium sulfate heptahydrate (3 cups) and magnesium chloride hexahydrate (5 cups) in enough purified freshwater to make 1 gallon total volume. There will likely be a precipitate that forms even if you fully dissolve both ingredients separately. That precipitate is calcium sulfate (calcium as an impurity in the magnesium chloride and sulfate from the Epsom salts). It is fine and appropriate to dose the precipitate along with the remainder of the fluid by shaking it up before dosing.
VS the "Sodium hydroxide version" from post 1 in this thread.
Thank you!
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
I've switched to this recipe from the original and All for reef, for the PH boost, its been great.
Im wondering how strong I can make the solutions. so ESV is 1.5, but I'm looking to make a 2x strength. is 3x or 4x possible? what are the solubility limits
There's no practical solution solubility limit for the alk part. It can be more solids than water. The calcum part is not far behind.
there may be usage reasons, however, to not concentrate it too much. You want the alk part to mix in fast. The more concentrated it is to start, the harder that is.
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,631
- Reaction score
- 64,091
I've been reading over these threads over the last few days. Thanks for the wealth of knowledge! I'd like to incorporate a 2-part solution that will raise my pH as high as possible to replace kalk in my ATO.
I saw this pH boost recipe in another thread. Could you help me to understand the difference in these two recipes, I'm having trouble combing through the threads to compare:
From the other thread:
Alk part
Add 283 grams of sodium hydroxide to 1 gallon of fresh water. It will get quite warm. Make sure it doesn't soften your container. This solution will contain about 1,900 meq/L of alkalinity (5,300 dKH). BE CAREFUL WITH THIS SOLUTION: IT HAS A pH ABOVE 14. Do not get it in your eyes or on your skin. Keep all reef chemicals, especially this alk part, in a way that children cannot access them.
Calcium part
Dissolve 500 grams (about 2 ½ cups) of calcium chloride dihydrate (such as Dowflake 77-80% calcium chloride or ESV calcium chloride; see below for substitutes and sources) in enough water to make 1 gallon of total volume. You can dissolve it in about ½ gallon of water, and then pour that into the 1 gallon container and fill it to the top with more freshwater. This solution has about 37,000 ppm calcium.
Magnesium part
Dissolve Epsom salts (magnesium sulfate heptahydrate (3 cups) and magnesium chloride hexahydrate (5 cups) in enough purified freshwater to make 1 gallon total volume. There will likely be a precipitate that forms even if you fully dissolve both ingredients separately. That precipitate is calcium sulfate (calcium as an impurity in the magnesium chloride and sulfate from the Epsom salts). It is fine and appropriate to dose the precipitate along with the remainder of the fluid by shaking it up before dosing.
VS the "Sodium hydroxide version" from post 1 in this thread.
Thank you!

Randy Recommended DIY Two Part Recipes
There are a whole lot of different DIY recipes around now, and I thought I lay out the differences and my recommendations. In all cases, food grade, USP (pharma) grade, or Analytical Reagent/ACS grades are all good choices for materials. Lower or ungraded materials may be fine, but its more...
 www.reef2reef.com
www.reef2reef.com
My recommendation is recipe C. A three part using Balling Part C for third part.
Two other recipes are a two part or a three part using sodium hydroxide. I'd personally use the three part described in post 1 of this thread:

Second New DIY Two Part Recipe with Higher pH Boost
In a previous thread, I posted a true two part DIY recipe: https://www.reef2reef.com/threads/new-diy-two-part-recipes-with-higher-ph-boost.344500/ But some folks may want to just swap the new ingredient into my 2/3 part recipe (as used by BRS, for example). Here's the original recipe link...
 www.reef2reef.com
www.reef2reef.com
- Joined
- Jul 12, 2019
- Messages
- 111
- Reaction score
- 98
I've done 4x concentration, the issue I ran into was it would settle in my sump like a gel. So I had to add powerhead to get some turbulence to properly mix.I've switched to this recipe from the original and All for reef, for the PH boost, its been great.
Im wondering how strong I can make the solutions. so ESV is 1.5, but I'm looking to make a 2x strength. is 3x or 4x possible? what are the solubility limits
Similar threads
- Replies
- 1
- Views
- 123
-
- AMS: Article
- Replies
- 61
- Views
- 4,428
- Replies
- 5
- Views
- 473
- Replies
- 8
- Views
- 165