What is your idea? Confirm what?idea: someone do a confirmational experiment, make my millennia
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Prime Does Not Remove Ammonia
- Thread starter Dan_P
- Start date
- Tagged users None
I truly would like to know about marine unassisted cycling more than any aspect of cycling in general. second to that is the ability or inability to starve a cycle proven to be ready/implanted on surfaces from an open topped common reef tank arrangement in someone's home.
for the unassisted cycle if someone would stack rocks in a bucket and keep it heated, circulated and topped off for three months and then do a basic oxidation test for ammonia I think its a big deal. that data is missing in the hobby within the direct confines of a reef tank and the dilution and typical flow and surface area positioning we use (several rocks stacked right in the middle of water contact)
Dan I swear I wont derail further past this just taking a shot while its here
I think the experiment should be two buckets, one dosed to .5 ppm for a light proofing
and then one dosed to 2 ppm just to chart how many days it takes to move back down, both these load tested after 90 days wait lead up. open topped but topped off, the room contaminants are the suspected feed input and non boiled water preps were the seeding bacteria input/as the hypethesis.
lastly, if someone is human-eyeing this vs Dan's light assessment method/something relatively objective/that devalues the findings but only if they fail
heh
but thank you for asking. I know you could find a way to test it well.
am seeking input on aspects of cycling there isn't a macna video on, or a post pattern set (though I think Steven started with a nice initial volley) or an article--and something that is always questioned by new cyclers.
end of non prime rant.
for the unassisted cycle if someone would stack rocks in a bucket and keep it heated, circulated and topped off for three months and then do a basic oxidation test for ammonia I think its a big deal. that data is missing in the hobby within the direct confines of a reef tank and the dilution and typical flow and surface area positioning we use (several rocks stacked right in the middle of water contact)
Dan I swear I wont derail further past this just taking a shot while its here
I think the experiment should be two buckets, one dosed to .5 ppm for a light proofing
and then one dosed to 2 ppm just to chart how many days it takes to move back down, both these load tested after 90 days wait lead up. open topped but topped off, the room contaminants are the suspected feed input and non boiled water preps were the seeding bacteria input/as the hypethesis.
lastly, if someone is human-eyeing this vs Dan's light assessment method/something relatively objective/that devalues the findings but only if they fail
heh
but thank you for asking. I know you could find a way to test it well.
am seeking input on aspects of cycling there isn't a macna video on, or a post pattern set (though I think Steven started with a nice initial volley) or an article--and something that is always questioned by new cyclers.
end of non prime rant.
Can you ask a concise question - or 2? i.e. question 1. Question 2. I have no clue what you are wanting - and I have (I think ) the ability to answer most questions - as do Dan and TarichaI truly would like to know about marine unassisted cycling more than any aspect of cycling in general. second to that is the ability or inability to starve a cycle proven to be ready/implanted on surfaces from an open topped common reef tank arrangement in someone's home.
for the unassisted cycle if someone would stack rocks in a bucket and keep it heated, circulated and topped off for three months and then do a basic oxidation test for ammonia I think its a big deal. that data is missing in the hobby within the direct confines of a reef tank and the dilution and typical flow and surface area positioning we use (several rocks stacked right in the middle of water contact)
Dan I swear I wont derail further past this just taking a shot while its here
I think the experiment should be two buckets, one dosed to .5 ppm for a light proofing
and then one dosed to 2 ppm just to chart how many days it takes.
lastly, if someone is human-eyeing this vs Dan's light assessment method/something relatively objective/that devalues the findings but only if they fail
heh
but thank you for asking. I know you could find a way to test it well.
am seeking input on aspects of cycling there isn't a macna video on, or a post pattern set (though I think Steven started with a nice initial volley) or an article--and something that is always questioned by new cyclers.
end of non prime rant.
- Joined
- Sep 21, 2018
- Messages
- 6,773
- Reaction score
- 7,248
We have a situation where the total ammonia test results can be ambiguous, e,g., low end API results seem to confuse users. API users always have 0.25 ppm, kinda. Are any hobby test kits going to give anything but false positives for 0.1 ppm. So thinking out loud, will the Spotte guidelines be getting people worried for nothing?While I wouldn't "panic", I do suggest folks take corrective action at those levels. I give my suggestions and reasoning here:
Ammonia Concentration Guidelines
Because ammonia's toxic effects appear at levels significantly below those that are acutely lethal (0.09 to 3.35 ppm NH3-N or 1.3 to 50 ppm total NH4-N at pH 8.2), and because some organisms in a reef aquarium may be more sensitive than the few organisms that have been carefully studied, it is prudent to err on the side of caution when deciding what concentrations of ammonia to allow in a reef aquarium or related system.
My suggestion is to take some sort of corrective action if the total ammonia rises above 0.1 ppm. This suggestion is also made by Stephen Spotte in his authoritative text, Captive Seawater Fishes.6 Values in excess of 0.25 ppm total ammonia may require immediate treatment, preferably involving removal of all delicate (ammonia sensitive) organisms from the water containing the ammonia. Some of the possible actions to take are detailed in the following sections listed below.
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,894
- Reaction score
- 64,327
We have a situation where the total ammonia test results can be ambiguous, e,g., low end API results seem to confuse users. API users always have 0.25 ppm, kinda. Are any hobby test kits going to give anything but false positives for 0.1 ppm. So thinking out loud, will the Spotte guidelines be getting people worried for nothing?
I agree that kit reliability is an issue, especially with the cheapest ones.
My strong feeling - based on doing perhaps 70 of these tests - is that people are doing them incorrectly - AND ITS EASY TO DO. For example - the guy that said - I held my finger over the vial as I mixed it, etc etc. I have to admit - I have no clue what the 'spotte' guidelines are - but without knowing - I will say - yes - they areWe have a situation where the total ammonia test results can be ambiguous, e,g., low end API results seem to confuse users. API users always have 0.25 ppm, kinda. Are any hobby test kits going to give anything but false positives for 0.1 ppm. So thinking out loud, will the Spotte guidelines be getting people worried for nothing?
- Joined
- Sep 21, 2018
- Messages
- 6,773
- Reaction score
- 7,248
Haha, let’s get off track. I am growing and testing BioSpira biofilms and I am not so sure they stay fully active when the ammonia is depleted. I need to do much more work to clarify this. Also, I am no longer certain about where the ammonia oxidizing activity is occurring during the first 2-4 weeks of cycling. We say the surface but my data suggests otherwise. I need much more data to straighten this out.I truly would like to know about marine unassisted cycling more than any aspect of cycling in general. second to that is the ability or inability to starve a cycle proven to be ready/implanted on surfaces from an open topped common reef tank arrangement in someone's home.
for the unassisted cycle if someone would stack rocks in a bucket and keep it heated, circulated and topped off for three months and then do a basic oxidation test for ammonia I think its a big deal. that data is missing in the hobby within the direct confines of a reef tank and the dilution and typical flow and surface area positioning we use (several rocks stacked right in the middle of water contact)
Dan I swear I wont derail further past this just taking a shot while its here
I think the experiment should be two buckets, one dosed to .5 ppm for a light proofing
and then one dosed to 2 ppm just to chart how many days it takes to move back down, both these load tested after 90 days wait lead up. open topped but topped off, the room contaminants are the suspected feed input and non boiled water preps were the seeding bacteria input/as the hypethesis.
lastly, if someone is human-eyeing this vs Dan's light assessment method/something relatively objective/that devalues the findings but only if they fail
heh
but thank you for asking. I know you could find a way to test it well.
am seeking input on aspects of cycling there isn't a macna video on, or a post pattern set (though I think Steven started with a nice initial volley) or an article--and something that is always questioned by new cyclers.
end of non prime rant.
Part of the problem involves (IMHO) - the ability of the average person to actually follow instructions. There are at least multiple examples where people do not - and get spurious results. There is no simple solution to this. My opinion - which some would feel is worthless-My strong feeling - based on doing perhaps 70 of these tests - is that people are doing them incorrectly - AND ITS EASY TO DO. For example - the guy that said - I held my finger over the vial as I mixed it, etc etc. I have to admit - I have no clue what the 'spotte' guidelines are - but without knowing - I will say - yes - they are
1. The test instructions are ambiguious
2. The people doing the tests do them incorrectly
3. The tests are inaccurate
my opinion is that this applies to API as well as others,
See the problem here - is that you are assuming that you are growing 'biospira biofilms'. No clue at all whether you are or not, But its a commentHaha, let’s get off track. I am growing and testing BioSpira biofilms and I am not so sure they stay fully active when the ammonia is depleted. I need to do much more work to clarify this. Also, I am no longer certain about where the ammonia oxidizing activity is occurring during the first 2-4 weeks of cycling. We say the surface but my data suggests otherwise. I need much more data to straighten this out.
And if you are - so what?See the problem here - is that you are assuming that you are growing 'biospira biofilms'. No clue at all whether you are or not, But its a comment
I think the question being asked is:Can you ask a concise question - or 2? i.e. question 1. Question 2. I have no clue what you are wanting - and I have (I think ) the ability to answer most questions - as do Dan and Taricha
Is the indigenous bacteria population in the room able kick start and develop sufficient nitrification capabilities in an otherwise untreated aquarium.
I think the answer is 'over time' yes.I think the question being asked is:
Is the indigenous bacteria population in the room able kick start and develop sufficient nitrification capabilities in an otherwise untreated aquarium.
This related to the ability to grow ' xxxx biofilms'. My guess is bacteria will compete, etc - there is no guarantee that biospira are growing on biofilms. Unless there is a reason to believe it
I thought I was talking about biospiraI think the question being asked is:
Is the indigenous bacteria population in the room able kick start and develop sufficient nitrification capabilities in an otherwise untreated aquarium.
And that's with no supplementation of ammonia/food from any sources?I think the answer is 'over time' yes.
If so, where does the colony gets their energy from?
Of course I think this is probably academic, not many people would be keen to do a fullu unassisted cycle nowadays, even if it works.
Oops. I should have made it clear I was refering to @brandon429 's post.I thought I was talking about biospira
By the way - I dont mean to dis- you - but if you could send a sample of your biofilm into aquabiomics - to verify what you are growing (unless you have another method). I'm just not sure what it means. 1. Perhaps biospira contains heterotrophs , etc etc. Who knows. I am going to make a proposal to R2R for funding for people who do experiments - which cost a bit of money and time - that they be funded - as is BRS.And if you are - so what?
J1a, it's not like the thread isn't right there for parsing and proof, give page 97 onward a read
Don't think I'm convinced easily by haphazard testing or not from doing it thirty years straight in various freshwater setups
Team to chat about it we should link up in multi chat i promised Dan I wouldn't redirect away from prime any further after my gratuitous first go which i appreciate so greatly because it might lead to furtherance of rare cycling info
Yes 100% unassisted wait based only (3 months in an open topped circulated heated topped off bucket reef test)
Don't think I'm convinced easily by haphazard testing or not from doing it thirty years straight in various freshwater setups
Team to chat about it we should link up in multi chat i promised Dan I wouldn't redirect away from prime any further after my gratuitous first go which i appreciate so greatly because it might lead to furtherance of rare cycling info
Yes 100% unassisted wait based only (3 months in an open topped circulated heated topped off bucket reef test)
- Joined
- May 22, 2016
- Messages
- 6,621
- Reaction score
- 10,212
The orange circle is the response of the ammonia sensing film to the Prime+ammonia solution, indicating no change to the final ammonia concentration.
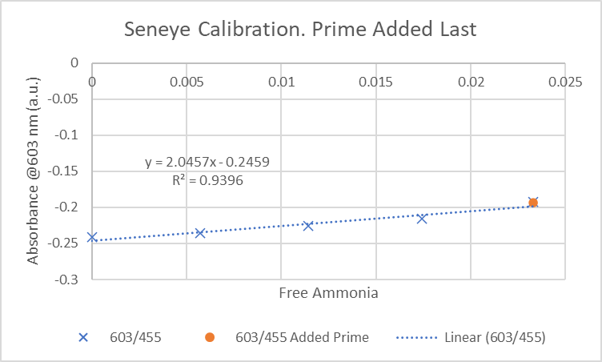
This is just one of dozens of experiments where I measured no ammonia removal.
Dan, could you zoom in on the Y-axis for me here?
Yes - @Dan_P tried that with (I cant remember) - but it required an extremely high pH - and high ammonia level like 8 ppm - which was outside the parameters that the Seneye measures accurately
To be clear, MnFish1 said this a few times in a few threads that we did this, but it isn't the case. I didn't use any seneye or seachem or any other film device to "measure" the absurdly high NH3 in the samples that was needed to kill amphipods. Of course those sensitive films are maxed out well before anything approaching that range.@taricha used some little saltwater crustacean.
I determined NH3 by careful stock addition and calculation / pH control. I plopped a seneye disk in each sample just as a check that no huge reduction of ammonia occurred. They all stayed very blue with or without Prime. That only says there was "lots" of NH3. The "measurement" part, was that amphipods stopped swimming normally at the same time frame and died in the same time frame with X ppm ammonia or X ppm of ammonia + a linearly scaled matching dose of Prime - that was the NH3 "measurement" part.
Yeah, at least ammonia coming from tap water chloramine. Prime will make the NH3 sensing films show clearly lower NH3 in the seachem films than they do if you dechlorinate with thiosulfate. i.e. The NH3 coming from the chloramine is significantly lowered by Prime....BUTInteresting thought. Does Prime reduce ammonia in freshwater? I wonder if @taricha looked this?
...if you measure pH you'll find that the reduced NH3 is due to lower pH from dechlorinating with Prime than with thiosulphate. And when you add a small amount of buffer to bring them to the same pH (8.2 when I did it), Prime disks color back up indistinguishable to the ones in the samples dechlorinated by thiosulphate. That is, simply correcting/controlling the pH makes prime indistinguishable from the zero effect of thiosulphate for removing the NH3 generated by dechlorinating tap water chloramine.
So yes Prime offers significant NH3 reduction in tap water by lowering pH. Buffer/correct pH to SW levels and the NH3 reduction disappears.
This was my experience from looking into it. I found no "live food" type organisms available that had published LC50 NH3 concentrations anywhere in the realm of plausible concentrations of interest in hobby systems. Not even ammonia - dosed cycling ones.Mysis are among them, but it takes high ammonia. The 48 h LC50 for 2 day old mysis is about 1 mg/L unionized (that is, free ) NH3. Ghost shrimp are even higher.
Talk about crazy and perfect timing! Neighbor just brought over a 4L bottle of prime that someone donated to her school lol. Seal unbroken id be more than willing to donate for further experiments.

Similar threads
- Replies
- 38
- Views
- 909
- Replies
- 2
- Views
- 119
- Replies
- 59
- Views
- 3,027
- Replies
- 8
- Views
- 185
New Posts
-
-
-
MACNA is Back !! Is anyone excited or will wait and see?
- Latest: vetteguy53081