It has been a lot of discussion and speculation why ORP drops directly after H2O2 have been added. In my aquarium - around 320 L - i always get a drop when I fill up my oxidator - it is always some uncatalyzed H2O2 that goes directly out into the water column when I fill up the container
It happens around 1 minute after the fill up (påfyllnad)
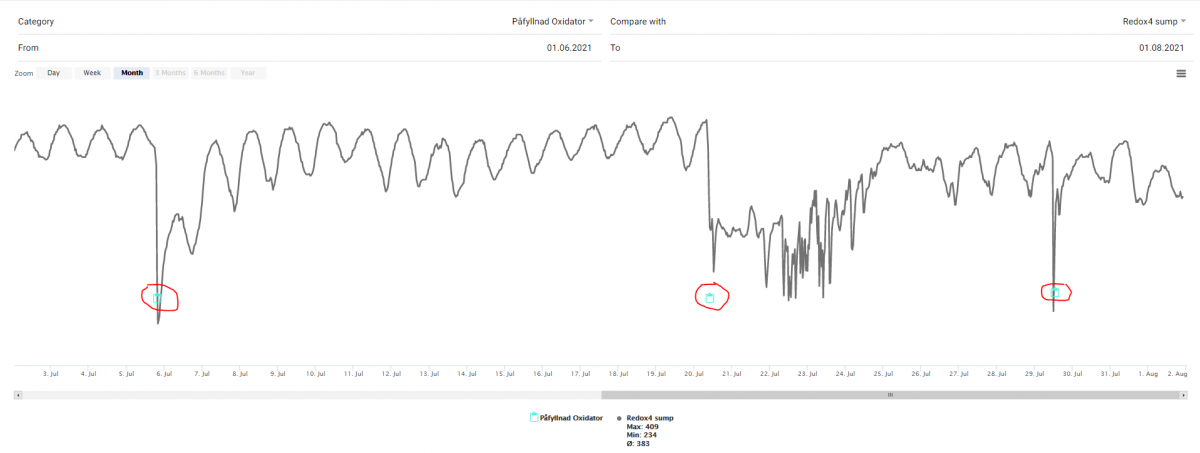
Possible reason for this had been suggested to be 1) The H2O2 breaks up cells 2) H2O2 can be both a reducer and oxidizer 3) false reading because of local reaction between the metallic part of the redox probe. 4) reactions in the saltwater.
My idea is to first start with 10 L RO water and 10 L tap water. Let them stand a while so it will be a steady reading. One redox electrode in each bucket. A small pump circulating each bucket. Sampling the ORP every 5 minutes in the beginning. After some days I will add 10 ml 12% H2O2 to each bucket and see whats happen.
If nothing drastic happens in the bucket with RO water - interaction of the measurements tool is unlikely. If something happens - we can´t exclude interaction off with the redox electrode (platina in contact with water)
If nothing happens in the tap water bucket - an indication that the ion strength is not enough to cause this. The tap water TDS value will be measured in both before the adding.
After this I will test with one bucket of fresh mixed saltwater and one containing 4 years old saltwater. This in order to get a hint if organics play a major roll in the ORP dip or not.
I will play around with a potato too - in all 4 buckets
I will post updates
Sincerely Lasse
It happens around 1 minute after the fill up (påfyllnad)
My idea is to first start with 10 L RO water and 10 L tap water. Let them stand a while so it will be a steady reading. One redox electrode in each bucket. A small pump circulating each bucket. Sampling the ORP every 5 minutes in the beginning. After some days I will add 10 ml 12% H2O2 to each bucket and see whats happen.
If nothing drastic happens in the bucket with RO water - interaction of the measurements tool is unlikely. If something happens - we can´t exclude interaction off with the redox electrode (platina in contact with water)
If nothing happens in the tap water bucket - an indication that the ion strength is not enough to cause this. The tap water TDS value will be measured in both before the adding.
After this I will test with one bucket of fresh mixed saltwater and one containing 4 years old saltwater. This in order to get a hint if organics play a major roll in the ORP dip or not.
I will play around with a potato too - in all 4 buckets
I will post updates
Sincerely Lasse

















